All Orders Ship Within 48Hrs. Industrial Mfg Experience. What are some real life examples of distillation? What is the difference between distillation and filtration? How to explain simple vs.
It is exemplified at its simplest when steam from a kettle becomes deposited as drops of distilled water on a cold surface. Initially, distillation is believed to have been used to make perfumes. Simple distillation is a procedure by which two liquids with different boiling points can be separated. However, before we begin a discussion of distillation , it would probably be beneficial to define the terms that describe the process and related properties.
Distillation was known to be used by the Babylonians of Mesopotamia. Fractional distillation works because the different liquids have different boiling points. The evaporation and subsequent collection of a liquid by condensation as a means of purification: the distillation of water. The extraction of the volatile components of a mixture by the condensation and collection of the vapors that are produced as the.
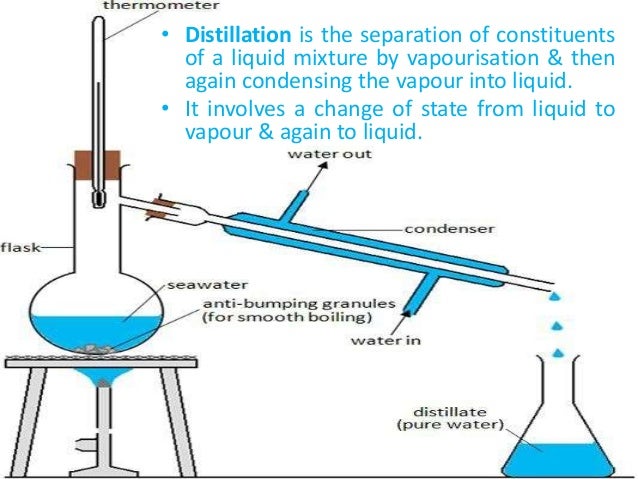
It involves a change of state, as of liquid to gas, and subsequent condensation condensation, in physics, change of a substance from the gaseous (vapor) to the liquid state (see states of matter). Separation exploits differences in boiling point, or volatility, between the components in the mixture. The successful application of distillation techniques depends on several factors.
The mixture is heated until one of the components boils (turns to a vapor). The vapor will contain a higher concentration of the more volatile component, so more of it will be condensed and removed from the system. The technique is widely used in industry, for example in the manufacture and purification of nitrogen, oxygen and the rare gases. When the liquids’ boiling points are very similar, however, separation by normal distillation becomes ineffective or impossible.
Koch Modular Process Systems , LLC. The atmospheric pressure measured in mmHg is the current atmospheric pressure in the laboratory. The RFD is a form of short path distillation that uses Ultra-High Vacuum levels to achieve molecular separation of target botanicals from crude oil and distill the remaining volatiles and terpenes.
A distillation apparatus is chemistry equipment commonly used to purify liquids and separate components in mixtures. Humans cannot drink saline water, but, saline water can be made into freshwater, for which there are many uses. The process is called desalination, and it is being used more and more around the world to provide people with needed freshwater. Read our guide to distilling to learn the primary ways liquor is made.
Our intro explains how alcohol is made in plain English. Water is distilled to remove impurities—for example, to remove salt from seawater. Air is distilled to separate its components, such as oxygen, which is used for medical applications, and helium, used for party balloons. For example, fresh water can be obtained from seawater (water that contains salts) by distillation.
Synonyms for distillation at Thesaurus. Find descriptive alternatives for distillation. A method called fractional distillation , or differential distillation , has been developed for certain applications, such as petroleum refining, because simple distillation is not efficient for separating liquids whose boiling points lie close to one another. Home Distiller Distilling to Making Your Own Essential oil, Moonshine, 3. There are many types of distillation columns, each designed to perform specific types of separations, and each design differs in terms of complexity.
In chemistry, azeotropic distillation is any chemical process, using which a liquid mixture containing azeotropes can be separated into its original components, by the addition of another substance (for instance, an entrainer), to enable distillation.
ليست هناك تعليقات:
إرسال تعليق
ملحوظة: يمكن لأعضاء المدونة فقط إرسال تعليق.